Guest Post by Dr. Robert Humphreys
 Global production of polystyrene (PS) and polyethylene terephthalate (PET) is gargantuan by any standard, reaching 14.3 million tonnes and 90.9 million tonnes (15.7 million tons and 100 million tons), respectively, in 2013 and growing at least as rapidly as population. Despite the numerous benefits to consumers, restaurants, food producers and sellers, supply chain logistics, and industrial manufacturers (see Figure 1, images taken from 1a, 1b, 1c, 1d, 1e, 1f), the ubiquity of waste PS and PET packaging along highways and beaches and in landfills led to a barrage of adverse publicity for these and other thermoplastics. In addition, like most thermoplastics, PS and PET are obvious targets for climate change concerns because they are petrochemically based. So, it should come as no surprise that huge, established markets and the sustainability movement, combined with the sometimes irrational exuberance of the early days of biofuels and renewable chemicals, made PS and PET irresistible targets for “sustainable” replacement technologies. Funding from the investment community, major corporations, and governments poured into start-ups focusing on sustainable versions of, or alternatives to, PS and PET. After over a decade of R&D and scale-up effort, countless, additional infusions of capital from investors (often with the requisite stock dilution), and numerous “partnerships” between start-ups and potential global customers, it seems appropriate to examine the state of sustainable PS and PET development and commercialization.
Global production of polystyrene (PS) and polyethylene terephthalate (PET) is gargantuan by any standard, reaching 14.3 million tonnes and 90.9 million tonnes (15.7 million tons and 100 million tons), respectively, in 2013 and growing at least as rapidly as population. Despite the numerous benefits to consumers, restaurants, food producers and sellers, supply chain logistics, and industrial manufacturers (see Figure 1, images taken from 1a, 1b, 1c, 1d, 1e, 1f), the ubiquity of waste PS and PET packaging along highways and beaches and in landfills led to a barrage of adverse publicity for these and other thermoplastics. In addition, like most thermoplastics, PS and PET are obvious targets for climate change concerns because they are petrochemically based. So, it should come as no surprise that huge, established markets and the sustainability movement, combined with the sometimes irrational exuberance of the early days of biofuels and renewable chemicals, made PS and PET irresistible targets for “sustainable” replacement technologies. Funding from the investment community, major corporations, and governments poured into start-ups focusing on sustainable versions of, or alternatives to, PS and PET. After over a decade of R&D and scale-up effort, countless, additional infusions of capital from investors (often with the requisite stock dilution), and numerous “partnerships” between start-ups and potential global customers, it seems appropriate to examine the state of sustainable PS and PET development and commercialization.
In this and the next several posts, we will examine some of the challenges that face any new technology that will compete with petrochemically derived polymers, the status of sustainable replacements for petrochemically-sourced PS and PET, and what may be required to achieve commercial success on a scale that matters substantively to sustainability.
Figure 1. Everyday products from polystyrene (PS) and polyethylene terephthalate (PET)
Efficient Use of Raw Materials: Improve or Die
The petroleum refining industry as we know it is well over a century old and has become remarkably efficient at converting the raw material, crude oil (Figure 2, image taken from 2a), into commercially valuable products.
Figure 2. Crude oil before refining
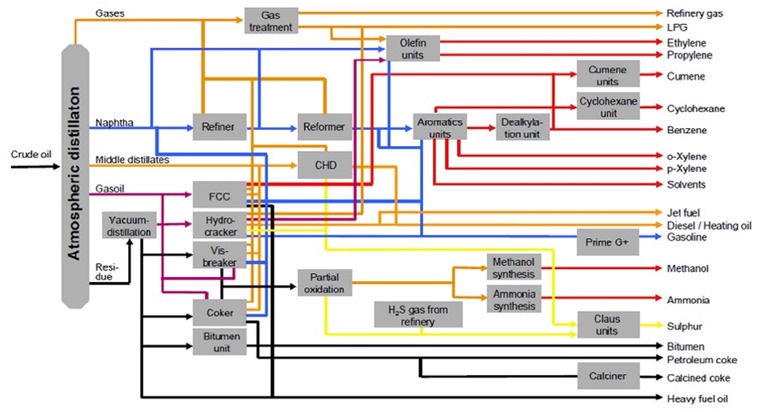
Essentially every drop of a barrel of crude oil is converted into products that can be sold, ranging from hydrocarbon gases (e.g. methane, ethane, propane, butane) to distillation “bottoms” (e.g. asphalt, tar, petroleum coke). In modern conversion and deep conversion refineries found in the USA (all US refineries are of the conversion or deep conversion type, see page 17, oil refining), much of the refinery complexity (see Figure 3, images taken from 3a, 3b) is devoted to “upgrading” crude oil to generate a larger percentage of higher value products from lower value “heavier” crudes (see page 6, Types of Crude, Economics of petroleum refining).
Figure 3. Schematic of a petrochemical refinery for production of fuels and petrochemicals.
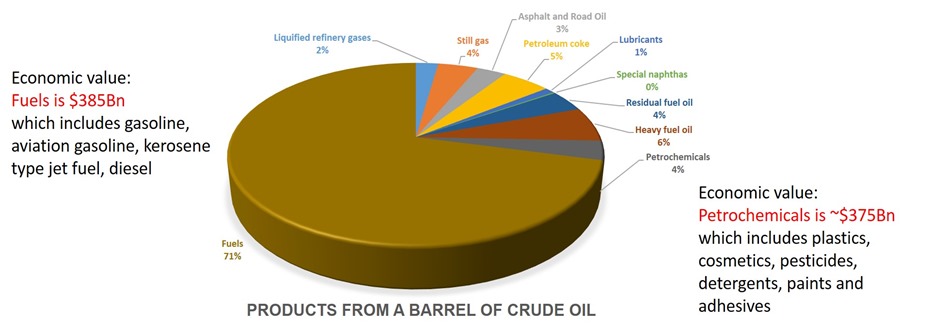
Upgrading includes removing sulfur and nitrogen (potential pollution sources), and “cracking” (breaking into smaller molecules with heat) and other processes for converting heavy, high-molecular-weight fractions (= very large molecules) of the crude oil into smaller molecules that can be used for gasoline, jet and diesel fuels, fuel oil, and petrochemicals. Petrochemicals, the highest value products from the refining process (see Figure 4), can be converted into an endless range of agricultural, industrial and consumer products that make modern life possible. Since oil refining and petrochemical production are commodity businesses, profitability over the long term depends on continuous improvement in efficiency of crude oil conversion to valuable products. It is this constant drive to upgrade crude for more efficient production of higher value products that results in the extremely complex, highly capital- intensive refineries we find in the USA today.
It is rare to find a member of the public, the media, or vocal critics of oil industry profitability who is aware of, let alone understands, the vast sums of R&D and capital investment required to build, run, maintain, and upgrade petroleum refineries and to discover and develop new sources of crude oil. In a similar vein, few understand how much they depend on petroleum for virtually every aspect of modern life and how the relentless drive for efficiency in production of petroleum and petroleum-derived products has made so many consumer goods affordable for everyone. Such naiveté is closely linked to unrealistic expectations about the ease of replacing crude oil with renewable sources of fuel, chemicals, and plastics upon which our economy is totally dependent.
This, then, is the world in which sustainable versions of, or sustainable alternatives to, PS and PET must compete if they are to challenge crude oil as the source of these ubiquitous materials. In the next post in this series, we will begin to examine what is required to replace petroleum as a source of these valuable polymers. In posts that follow, we will explain some of the challenges facing renewable technologies for PS and PET and will make some predictions about which technologies might win and why.
Figure 4. Products from a barrel of crude oil: why petrochemicals are so important
Source: www.eia.gov/dnav/pet/pet_sum_sna_d_nus_mbbl_m_cur.htm



Leave a Reply