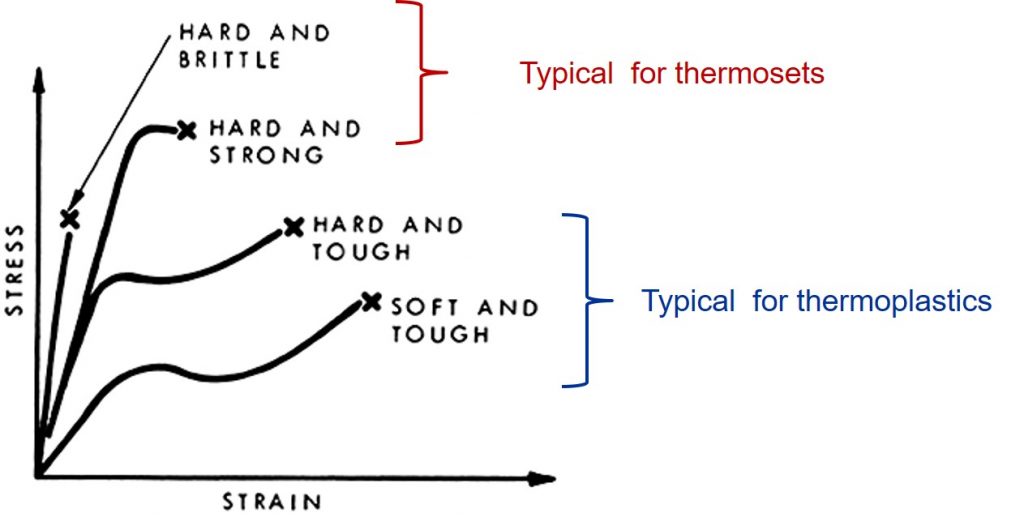
A previous post described the tensile testing of polymers. The tensile test typically measures the load extension curve (stress-strain as seen above) until the sample breaks. The toughness of the polymer can be estimated by the area under the stress strain curve. The curves for the thermosets have a high modulus (steep slope in the initial linear region), but have little elongation before they break. In contrast the thermoplastic materials can have a lower modulus (the soft and tough above) but much longer elongation. A logical question is why do the thermoplastics have so much elongation?
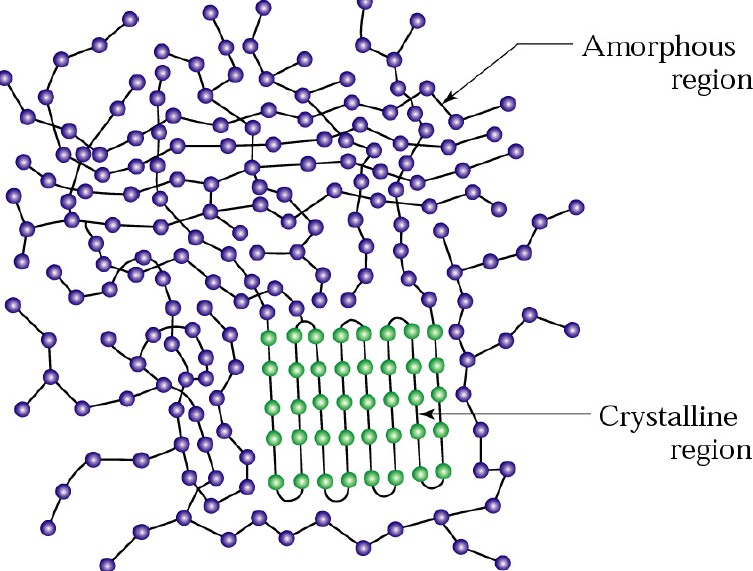
First, let’s examine the molecular structure of a simple thermoplastic (for example polyethylene). Polyethylene is a semi-crystalline polymer and contains both amorphous regions (the blue chains in Figure 1) and highly crystalline domains (the green chains in Figure1). The amorphous regions for semi-crystalline polymers can be either very large (low degree of crystallinity) or for highly crystalline polymers, the amorphous region is relatively small. Polyethylene has typically a high degree of crystallinity. For polyethylene, the crystalline domains are tightly packed in lamellae structures (1).
Figure 1. Schematic for a semi-crystalline high molecular weight polymer
The actual deformation of the polymer chains is very complex during tensile testing, but there have been some simplistic models to help understand the deformation mechanisms during tensile elongation. To begin, a semi-crystalline polymer has hard lamellae surrounded by amorphous chains. The application of a tensile load causes the deformation of the polymer lamellae along with the amorphous regions. A basic conceptual model was developed by JM Schultz (2) and is shown schematically in Figure 2.
Figure 2. Conceptual model of stretching mechanism for partially crystalline polymers (from reference 2)
In Figure 2a, prior to the application of the tensile load, the crystalline lamellae are closely connected by the amorphous regions between the tightly packed polymer chains. As the semi-crystalline polymer begins to deform, the stress causes the chains in the amorphous region to elongate and uncoil (b and c). At higher strains, the stress is transferred from the amorphous chains to the lamellae causing the crystalline lamellae to become oriented in the direction of the applied stress (d). Parts of the lamellae can break apart forming smaller fold blocks. With further levels of large elongations, the fold blocks continue to deform in the direction of the applied stress and the amorphous regions become highly elongated resulting in a fibrillar type structure.
Notice in the stress strain curve, the thermosets do not exhibit the increased load past the yield point. After the yield point, thermoplastics can strain harden. The mechanism of strain hardening is caused by the orientation of the crystalline chain blocks as seen in Figure 2d. As the amorphous chains and crystalline lamellae align in the direction of the applied stress (at 2e), the load bearing ability of the polymers increases. Using this phenomenon, polymer filaments can be made with very high tensile strength. A commercial product is Honeywell Spectra fiber, which is ultra-highly oriented polyethylene made using a patented gel-spinning process. It is stronger than steel and 40 percent stronger than aramid fiber.
What about thermosets? A schematic of a crosslinked thermoset is shown in Figure 3.
Figure 3. Schematic of crosslinked thermoset polymer
As discussed in many previous posts, chemical crosslinks are useful for producing fully cured thermosets with high modulus and good thermal stability. Under an applied tensile load, the amorphous polymer chains between the crosslinks can deform in an analogous manner to the amorphous chains in semi-crystalline polymers. There is a major difference. The chemical crosslinks prevent large polymer chain motion resulting in stress strain curves with very little deformation (low values of strain). The amount of crosslinking (crosslink density) and the molecular weight between the crosslinks governs the tensile stress-strain curves. At high loads, the crosslinked polymers chains cannot easily deform under the applied stress leading to both a high modulus, but low elongation to break. This is why thermosets while having very high moduli, are typically brittle (i.e have low elongations at break). Suitable tailoring of the crosslink density and increasing the flexibility of the chains between crosslinks can lead to the strong and hard curve above. This type of thermoset exhibits both the high modulus, high tensile strength and some ductility (larger elongation at break).
The next post will discuss fracture toughness and an introduction to fracture toughness testing.
References
- Eisele, U., Introduction to Polymer Physics, pages 79-86 Springer-Verlag, Berlin (1990)
- Schultz, J., Polymer Material Science, Prentice-Hall, Englewood Cliffs, New Jersey (1974)



Leave a Reply